For the vast majority of the history of human use of psychedelics, they have been seen as something sacred, a gift from God or mother nature herself. Ayahuasca, which contains DMT, was given out by shamans in the Amazon as a tea. It was a spiritual experience that would purge you of your negative energy and reveal the true nature of the universe to you.
The nature of psychedelics and the way people interact with them changed drastically in the 20th century. On November 16, 1938, Albert Hoffman first synthesized lysergic acid diethylamide (LSD), and 5 years later discovered its psychoactive nature. From then on, psychedelics no longer came exclusively from nature, and so some of the mystery behind their power slipped away. LSD became a favourite of both the counterculture youth, who thought it would free people’s minds, and the CIA, who hoped they could use it to control minds. And then, by 1968, psychedelics were illegal in the US.
In the following decades, psychedelic enthusiasts went a number of directions. Some continued to focus on the lab-made psychedelic LSD, while many began to search for psilocybe mushrooms themselves. Those who went back to searching for mushrooms in forests rediscovered the natural spirit that had once been an integral part of the psilocybin experience.

As psychedelic therapy companies have become the talk of the town, most have gone with a hybrid of the natural and artificial approach by using synthetic psilocybin. There are a few reasons for this – better control of dosage and psychedelic duration – but the main reason most psychedelic companies are choosing synthetic psilocybin over its natural counterpart is the same reason so many of these companies are based in Canada: patents. In Canada, you can’t patent a natural compound, but you can patent a slight variation on psilocybin that you produce synthetically.
But there are some who aren’t following the mainstream approach to the letter, like Filament Health (FH.NE), which is taking a different approach to the nature and scientific balance, and is showing that not being able to patent psilocybin isn’t such a big problem, because you can patent the extraction process.
Just last week, Filament received the first-ever psilocybin extraction and standardization patent for a public company by the Canadian Intellectual Property Office. According to the press release, the patent “describes essential technology for transforming variable psychedelic raw materials into pharmaceutical-grade standardized extracts.”
“We are proud to lead the industry with this first issued natural extraction patent. This approval represents important progress in the development of our intellectual property,” stated Benjamin Lightburn, CEO of Filament. “At Filament Health, we know that nature is a valuable source of medicine, but that certain technologies are necessary to bring natural products up to a pharmaceutical grade—most importantly, through standardization.”
Whatever you think about the various merits of going with the extraction of natural psilocybin versus synthesizing psilocybin, it’s should raise an eyebrow that one side of the divide has a virtual monopoly on the public markets and conversation at this point. Think of how many companies are using synthetic psilocybin: COMPASS Pathways (CMPS.Q), Mindmed (MNMD.Q), Field Trip Health (FTRP.T), Cybin (CYBN.NE), just to name a few. Those four companies have a combined market cap of $3.5 billion, and they are far from the only four companies in the synthetic psilocybin game. It’s safe to say, it’s a crowded lane. But Filament is out here in a lane of their own, flying ahead.

It’s worth noting that, although there are a near-endless number of synthetic psilocybin formulas, as companies can keep patent slight variations, there are only so many ways to extract natural psilocybin. That’s why Filament getting the first psilocybin extraction and standardization patent for a public company by the Canadian Intellectual Property Office is so important: there are only so many ways to do it, and Filament has one of them patented. Sure, COMPASS Pathways has some valuable IP in their COMP360 patents, but forever going forward they will be dealing with competitors with their own synthetic variations. Whichever synthetic psilocybin company is the first to have their compound approved will certainly have an advantage, but they won’t have a deep moat, because someone could, at any point, create a better, cheaper synthetic psilocybin compound.
But in the case of natural psilocybin, because there are only so many ways to extract psilocybin, being the first company with a patent in this sector gives them an advantage. Filament had their pick of the litter when deciding which of the finite ways to perform extraction they thought was most promising. Any subsequent competitors they do have will be choosing from an increasingly small menu of options on how to commercialize natural psilocybin. Getting there first on the natural extraction side of the psilocybin extraction business provides a deep moat.
There are five steps involved in the commercialization of natural psilocybin: propagation, extraction, purification, standardization, and distribution. Propagation involves growing the actual mushrooms, finding the best conditions, methods, genetic lines, etc. that will lead to the best quality psilocybin. Extraction consists of removing psilocybin, psilocin, baeocystin, and any other desired part of the mushroom. During purification, undesirable elements are removed, increasing the safety of the compound. Standardization results in consistent dosing, an important part of preparing the compound for clinical trials. And distribution comprises finding the best method to https://e4njohordzs.exactdn.com/wp-content/uploads/2021/10/tnw8sVO3j-2.pngister the compound, for instance, oral capsules, inhalation, or intravenously, to name a few methods. Of these steps, extraction and purification seem to offer the most fruitful ground for patents that provide early companies the most advantage.

Another noteworthy thing about Filament is how far along they are compared to how well known they are. There’s a good chance that many of you are just now hearing about them for the first time, but they already have a 3,500 square foot Good Manufacturing Practice (GMP) manufacturing, research and development facility on the British Columbia Institute of Technology (BCIT) campus. They also have their Health Canada Dealer’s License (#6-1289) permit allowing the propagation, extraction, and production and distribution and sale. That’s right, they’ve got a licensed shroom lab, with a license amendment submitted to include all controlled natural psychedelics, including DMT, mescaline, and harmaline. And they’ve still got $5.6 million in cash on hand on top of everything else.
Their dealer’s license has enabled them to propagate psychedelic plants, conduct genetic research, perform extraction procedures, run in-house trials, and distribute IP and drug candidates. Their PEX010, a proprietary psilocybin formulation, is already at Phase II trials, and their PEX020 and PEX030 non-psychedelic mushrooms are IRB approved and are in Phase I. All this by a company that listed on the NEO exchange less than two months ago, at a time when the company was already producing pharmaceutical-grade natural psilocybin.
At the time of the listing, Lightburn noted:
“Listing on NEO, a senior exchange with high governance requirements, helps to establish Filament as a leader in the psychedelics industry. NEO has proven to be a strong and supportive partner to innovative companies like Filament. The capital access made possible by this going-public transaction allows us to further our mission of getting safe, natural psychedelics into the hands of everyone who needs them, as soon as possible.”
While many psychedelic companies try to make as big a splash as possible, Filament has been purposefully flying under the radar. Instead of trying to drum public attention to grab as much money as possible, they operated in a stealth mode. They built much of the company before going public, and instead of going on wide roadshows, trying to land big institutional investors who are only in it to make a few bucks, they raised money from people who want to see the company succeed.
They also have a tight cap structure, because while they do have 164.7 million shares outstanding, a large portion are owned by company insiders who want to see the company grow, instead of just cashing out at the first opportunity. There are also only 15.6 million options and 9.3 million warrants issued (2 million of which are performance-based), also attesting to the tightness of their capital structure.
So how were they able to get this far without creating a huge media buzz or taking barrels of cash from big players? Because the founders are committed to the company and have experience building things. Ben Lightburn, Lisa Ranken, Filament’s COO, and Ryan Moss, head of R&D, all worked at Mazza Innovation, which was acquired by Sensient Technologies in 2018 for $26 million, after which they eventually left and started Filament. The money from this acquisition made it possible for, following incorporation, Ben Lightburn to fund the company, meaning they didn’t need those early rounds of funding where a just incorporated company has to sell shares for very little. Management is clearly committed to the company, and isn’t just looking for a way to cash out quickly.
But the commitment isn’t just about Filament; they believe in psychedelics in general, which is why they’ve created the Filament Foundation. Filament Founders have pledged 10% of their shares to a foundation which will help fund key actors and key initiatives to help the global psychedelics ecosystem thrive. The board is majority controlled by outsiders (3/5 members are not involved in Filament) and is committed to transparency. Between self-funding the company in the initial phase and the Filament foundation, it seems clear that those involved in Filament are deeply invested in the company’s success and the psychedelics industry at large.
If you want to hear Filament will be hosting a virtual investor event this Thursday, on August 12, 2021 at 2pm EDT, which you can register for online.
“We are incredibly proud of our progress thus far,” stated Lightburn. “We are currently manufacturing GMP pharmaceutical-grade natural psilocybin extracts, and will soon begin the first ever FDA-approved clinical trials of natural psilocybin. We look forward to discussing this, and other exciting updates, during our virtual event.”
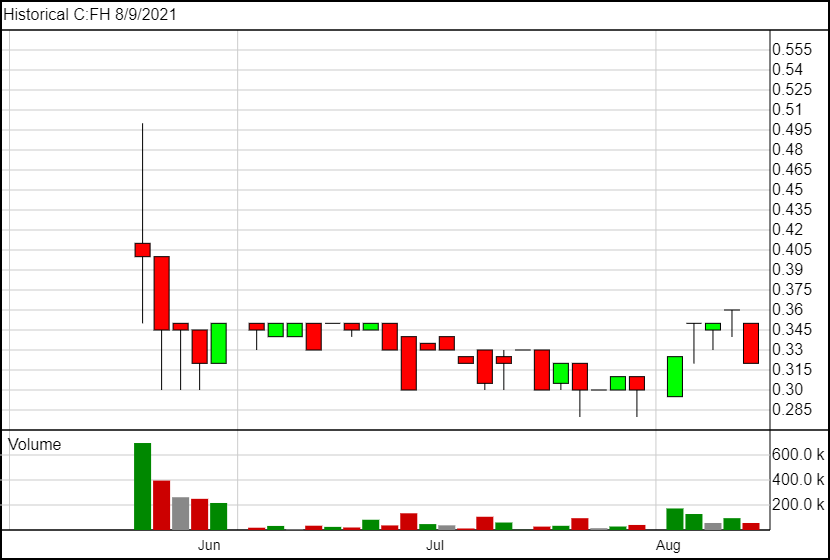
Today on August 9th, Filament shares are currently trading at $0.32, and have traded between $0.28 and $0.50 since going public.
Full disclosure: Filament Health is an Equity Guru marketing client.