Filament Health (FH.NE) is developing a pipeline of natural psychedelics products to treat mental health problems like depression and anxiety.
August 3, 2021, FH received a patent for the technology used to transform psychedelic raw materials, like psilocybin-bearing mushroom, into pharma-grade extracts.
“This approval represents important progress in the development of our IP,” stated Benjamin Lightburn, founder and CEO of Filament, “We know that nature is a valuable source of medicine, and that certain technologies are necessary to bring natural products up to a pharmaceutical grade.”
The 2020 Global Anxiety Disorder & Depression Treatment Market is about USD $16 billion. The market is forecast to reach USD $19.81 Billion by 2028, registering a CAGR of 2.4% over the forecast period.
Revenue growth is driven by increasing awareness about the tragic consequences of mental health disorders and the rising demand to treat them.

“Anxiety disorders and depression, are among the most prevalent mental health conditions,” states BioSpace, “Though they are less visible than schizophrenia and bipolar disorder, they can be just as disabling”.
In the US, recent estimates show 16 million adults had an episode of major depression in the course of a year.
“Filament has the capabilities to experiment and come up with interesting new compounds, extraction methods, and potential future products that probably go far beyond whatever is being discussed in mainstream psychedelics today,” wrote Equity Guru’s Taylor Gavinchuk on July 30, 2021, “We are only scraping the surface with fungi research.”
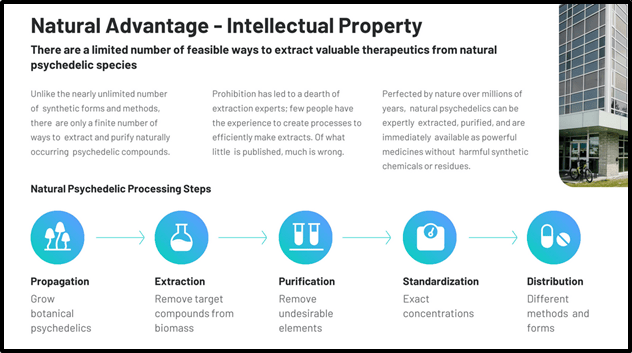
“With only around 100,000 species described, out of an estimated 5.1 million, fungi make ideal candidates for bioprospecting or extracting useful compounds for pharmaceuticals and other things from nature. And there are over 200 species of mushrooms that produce psilocybin. The possibilities are endless.
The company operates a 3,500 sq/ft lab just outside of Vancouver, BC that has the capacity to produce 25,000 therapeutic doses of psilocybin per year. Their in-house operations include propagation, extraction, research, and manufacturing using patent-pending technologies.” – End of Taylor.

In this June 22, 2021 video, Filament CEO Ben Lightburn describes Filament’s business objectives.
“Our mission is to see safe, natural psychedelics in the hands of everyone who needs them as soon as possible,” stated Lightburn, “The reason for starting Filament was the opportunity to bring together my experience and the experience of some former colleagues of mine which has been, throughout our careers, mostly in this area of botanical extraction.”
Lightburn and his team have developed a suite of technologies necessary to create pharmaceutical grade, natural psychoactive extracts. FH takes those extracts and puts them into FDA registered natural psychedelic clinical trials.
“For the first time people will have a choice to take a synthetic product, or a natural product,” added Lightburn, “We are excited to see is if naturally extracted compounds can provide even more benefits than synthetic.”
Lightburn calls the chemical synthesis processes “highly inefficient.”
“We’re taking psychedelics back to its roots and looking at the original natural sources, developing the technologies to put them in a safe and standardized preparation for a pharmaceutical clinical trials,” stated Lightburn, “And then running those clinical trials in partnership with the University of California San Francisco, in their leading psychedelic research group.”
In this June 29, 2021 TraderTV interview Lightburn addresses the “perceived investment downside” of a natural product over a synthetic product.
“You’ll hear synthetically focused companies say, ‘Natural is too expensive, it’s too hard to standardize, it’s not GMP, the FDA will never approve it, you can’t get IP protection’”, stated Lightburn.
“While there is a small element of truth in all of those things, in aggregate we believe that those beliefs are more or less falsely-held,” added Lightburn.
Lightburn points out that the largest selling cancer drug of all time was actually a natural extract, now sold under the brand name of Taxol.
Regarding IP protection for naturally occurring molecules, Lightburn admits that it’s not possible to get a patent on the molecule itself. However, you can patent processes that can turn raw natural biomass into a pharmaceutical grade extract.
“We think that the natural approach provides better opportunities for IP protection than synthetic products,” added Lightburn.
Consumer demand for organic products in general is surging.
“The North American organic sector posted a banner year in 2019, with organic sales in the food and non-food markets totaling a record $55.1 billion, up a solid 5% from the previous year”.
There hasn’t been a lot of comparative research into consumers of natural foods and natural drugs – but it seems likely that there is some overlap.
“How many mg for a “life changing trip”? Goal is to remove anxiety and become more self confident,” asks one mushroom enthusiast on a reddit forum.
“I’m microdosing psilocybin truffles at the moment taking 0.3mg every 3 days and the effects are great,” explained the reddit user, “Could 1 good trip fix underlying issues? Would appreciate guidance on how much and also what’s the expected length of it?”
Anxiety disorders and depression affect up to 13% of people in North America, making it the most common group of mental diseases.
The FDA has twice designated psilocybin as a “breakthrough therapy” for the treatment of severe and treatment-resistant depression.
“Anxiety disorders can be blamed for lower productivity, higher morbidity and mortality rates, and an increase in alcohol and drug usage in a broad segment of the population,” reports BioSpace, “Therefore to treat such problems, increased attention is being paid to anxiety disorder and depression treatment.”
“Over 40% of all new pharmaceutical products are discovered in the natural world,” concluded Lightburn.
Full Disclosure: Filament is an Equity Guru marketing client.