Cybin’s (CYBN.NE) subsidiary Adelia Therapeutics completed earnout milestones originally set in a contribution agreement for the period of January 1, 2021, following Cybin’s acquisition of Adelia.
There are a handful of science-heavy specifics to this development that are going to need some unpacking, but essentially the deal was about providing proof of concept for the manipulation of certain chemicals at an atomic level, so they could chase an investigational new drug designation from the FDA.
Here’s the heavy science from the press release.
“The achievement includes API Synthesis and optimization to demonstrate that two or more deuterated tryptamines show significant in vivo modifications of PK consistent with “Proof of Concept”, nomination of two deuterated candidates for full IND enabling studies, and completion of a certain API Manufacturing Contract.”
First, API Synthesis is short for Active Pharmaceutical Ingredient, and it’s defined by the Food and Drug Administration (FDA) as any substance or mixture of substances used as an active ingredient in drug manufacturing. Specialized expertise is required to synthesis these molecules. Secondarily, a deuterated drug is a small molecule medicinal product wherein the hydrogen atoms in the drug molecule have been replaced by the isotope, deuterium.
So these tryptamines, themselves metabolites of the amino-acid, tryptophan (best known for being the chemical in turkey meat that puts out grammas lights after Thanksgiving dinner), have had their hydrogen atoms removed and replace with deuterium. The idea behind it is that it can significant lower rates of metabolism, and therefore deliver a longer half-life. In this case, it’s produced pharmacokinetic effects on the body enough to prove that there’s potential for this to be a marketable product. Hence, the experiment worked and now they’re going to chase investigational new drug specification.
What is Cybin anyway?
Cybin is a biotech company working on developing drug discovery platforms, drug delivery systems, novel formulations, and treatment regimens for psychiatric disorders, using psychedelic therapeutics. Adelia is their subsidiary, and its aims include the development of medicinal psychedelics with improved dosing efficiency and the overall aim of meeting unmet medical needs. The strategic involves chemical alterations of tryptoamine derivatives which modify what they do to the body but without sacrificing their potential for therapeutic uses.
Their approaches minimize variables between patents by producing better controls for drug metabolism (the deuterium does this) without losing drug efficiency. Therefore, they can produce more predictable results and happier patients.
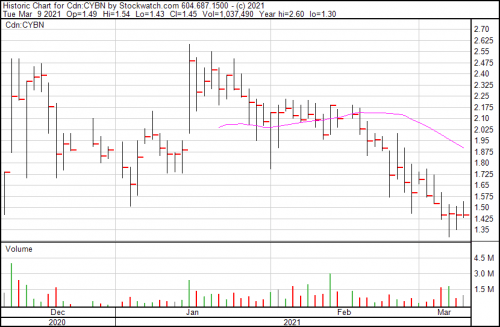
Shares were flat today and Cybin closed at $1.45.
—Joseph Morton
Full disclosure: Cybin is an equity guru marketing client.