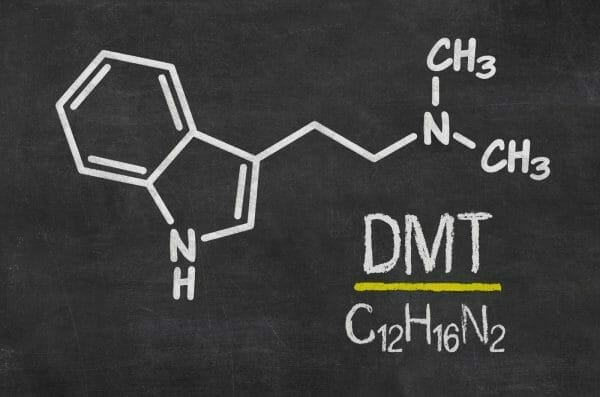
Algernon Pharmaceuticals (AGN.C) awarded the contract for the synthesis and manufacture of the psychoactive ingredient and finished product for its AP-188 DMT project to Canadian-based Dalton Pharma Services today, according to a press release.
Dalton has jumped through all the regulatory hoops as far as Health Canada, the US FDA and good manufacturing practices certification are concerned, and are a class 4 controlled substance licensed manufacturer with experience in synthesizing psilocybin and tryptamine derivatives. What they do is provide integrated chemistry, drug development and manufacturing services to the pharmaceutical and biotech industries.
“We are very pleased to have appointed Dalton Pharma Services to synthesize our GMP DMT supply. Based on positive pre-clinical data, the Company believes that DMT may help treat patients who have suffered from a stroke, which is one of the most devastating injuries a human being can experience,” said Christopher J. Moreau, CEO of Algernon Pharmaceuticals.
Algernon’s business model surrounds the idea of rooting through medicine cabinets and finding drugs used for one ailment, like Viagra for example, which in addition to making old men hotter than Phoenix in July, also has uses for pulmonary hypertension, or a condition involving increased blood pressure in the arteries of the lungs. The company then moves them swiftly and safely through new human trials and chases regulatory approval before shunting them off to market.
The work on synthesizing DMT has already started, and the company will soon file its pre-investigational new drug meeting request with the U.S. FDA, and get back to work on planning its Phase 1 and Phase 2 clinical studies.
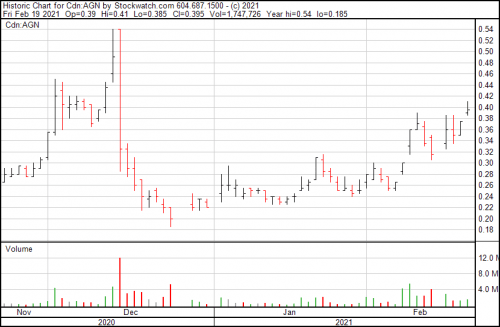
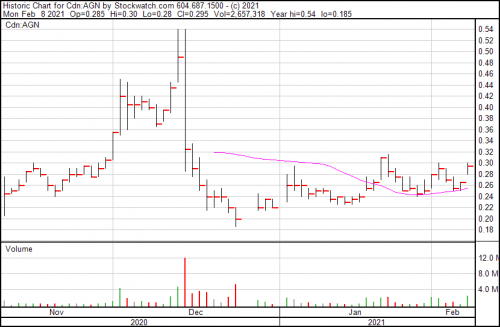
Algernon’s up $0.015, continuing a steady incline since late January, and bringing their share price to $0.39.
—Joseph Morton