Revive Therapeutics (RVV.C) announced they are entering into an exclusive license agreement with Puerto Rico Science Technology and Research Trust (PRSTRT) for the intellectual property titled ‘Biologically Active Ganoderma Lucidum Compounds and Synthesis of Anticancer Derivatives; Ergosterol Peroxide Probes for Cellular Localization’.
Yes, I know that was a lot of big words, but stick with me.
The PRSTRT (or just ‘the trust’), who represent Universidad Central del Caribe (UCC) and St. Jude Children’s Research Hospital, is allowing Revive to use its IP, which was developed by St. Jude and UCC. I’m gonna break down some of those giant words I used earlier. First, Ganoderma is a genus of mushroom, and Ganoderma lucidum is a type of mushroom, often confused for its cousin, Ganoderma lingzhi, a mushroom used in traditional Chinese medicine nicknamed ‘red reishi’. Ganoderma mushrooms are often believed to have anti-cancer properties, but this has not been definitively proven.
Second, Ergosterol Peroxide is a steroid derivative isolated in a number of sources, including fungi, specifically Ganoderma lucidum. It has been reported to exhibit several positive health outcomes, including antitumor properties, in vitro. Research has revealed significant co-localisation effects from Ergosterol Peroxide Probes.
Okay, that’s the two most uncommon terms out of the way, now on to the simpler stuff. A cellular localization process where a substance or cellular entity, such as a protein complex or organelle, is transported to/maintained in a specific location within a cell, including the localization of substances or cellular entities to the cell membrane. Biologically active is just a pharmacological term used to describe the beneficial or adverse effects of a compound.
Put it all together, and what do you get? The IP Revive is licensing is using extracts from a mushroom which has a long history of medicinal use in China to target cancer. Although Ganoderma has long been a popular medicine, there is not very much modern, western research to back up its health claims, although there have been a few studies that have shown some favourable results, including one that stated “the combined studies indicate that EP [Ergosterol Peroxide] from Ganoderma lucidum extract is a promising molecular scaffold for further exploration as an anti-cancer agent.”
“We are very excited about the new addition to our clinical-stage product pipeline as part of our long-term strategy of unlocking the therapeutic potential of medicinal mushrooms such as psilocybin and Ganoderma lucidum to treat mental health, substance abuse and aggressive cancers. Our intention is to build off from the research conducted at St. Jude Children’s Research Hospital and UCC by advancing towards FDA IND-enabling studies to allow for human clinical studies,” commented Michael Frank, CEO of Revive Therapeutics.
The agreement will give Revive the exclusive worldwide development and commercial rights to the patent for the Ganoderma Lucidum Extract (GLE) treatment. Revive agreed to future payments to the trust based on clinical trial and revenue milestones which are pursuant to industry standards.
The GLE treatment is primarily targeted at cancer, although GLE can also help treat and prevent hypertension and is effective against immunological disorders. Research has also suggested that GLE selectively inhibits breast cancer viability, induces apoptosis, reduces invasion, regulates key signaling molecules, and reduces tumor size by ~50% in mice xenografts.
In the press release, Revive wrote “The licensed invention elucidated the chemical structure of the most abundant chemical constituents of GLE and determined their efficacy in different aggressive breast cancer models. Several natural compounds were identified and three showed significant anticancer activity (ergosterol, 5,6-dehydroergosterol, and ergoesterol peroxide). In particular, one of these compounds, ergoesterol peroxide, exerts selective effects on cancer cell viability, similar to the effects produced when using whole mushroom extract GLE, suggesting an ample therapeutic window. Ergoesterol peroxide displays anti-proliferative effects through G1 phase cell cycle arrest, apoptosis induction via caspase 3/7 activation and PARP cleavage, decreases migration and invasion while inhibiting the expression of total AKT1, AKT2, BCL-XL, Cyclin D1 and c-Myc, induces ROS formation, and inhibits tumor growth in vivo. This technology further includes ergosterol peroxide chemical probes for in vitro anticancer evaluation, live cell studies, and proteomic profiling.”
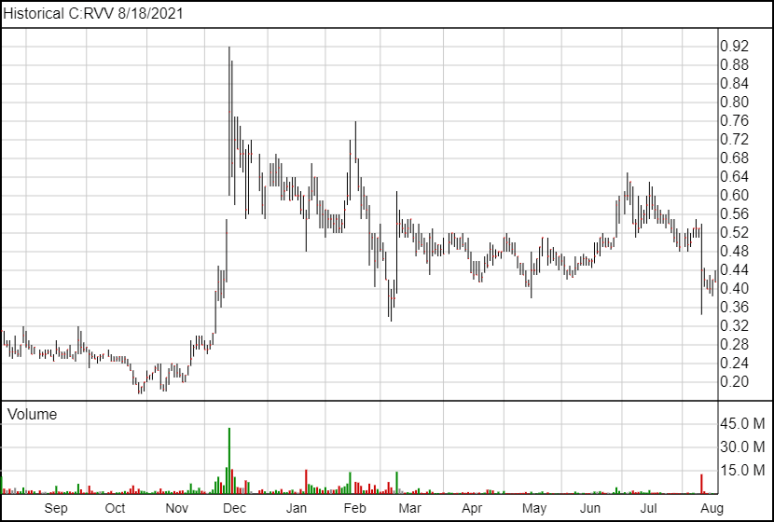
Following the news, Revive shares are up half a cent and are trading at $0.425.
Full disclosure: Revive Therapeutics are an Equity Guru marketing client.