NanoVibronix (NAOV.Q) announced today that it intends to enter the Over-the-Counter (OTC) pain relief market with the introduction of PainShield RELIEF™, a non-prescription ultrasound therapy device that delivers fast pain relief for nerve and soft tissue damage.
“Entering the OTC pain relief market with our newest product in the PainShield family significantly expands our total addressable market and opens up new opportunities for growth through several additional revenue streams. Those suffering from pain will have ready access to the device with no need for clinician prescription. Plans are under way for an official launch in the U.S. and select parts of the world…The company expects widespread availability by early to mid-2022,” said Brian Murphy, Chief Executive Officer of NanoVibronix Inc.
As previously mentioned, PainShield RELIEF™ is the latest member of NanoVibronix’s PainShield® family. PainShield® utilizes the Company’s patented low intensity surface acoustic wave (SAW) technology to provide pain relief. Using an adhesive patch and transducer, PainShield® is able to deliver ultrasonic energy to relieve pain and expedite soft tissue healing. If you’re blissfully ignorant like me, you were probably unaware that ultrasounds were useful for anything outside of helping suburban families plan for outrageous gender reveal parties. With that being said, let’s talk about what an ultrasound is. An ultrasound is a diagnostic imaging method that uses high-frequency sound waves to produce images of structures within our bodies. Similar to how PainShield® utilizes an adhesive patch, most ultrasound examinations are conducted by placing devices outside of the body.
With this in mind, NanoVibronix’s PainShield® uses these same sound waves to deliver localized pain relief and induce soft tissue healing in a targeted area, while keeping the level of ultrasound energy at a safe and consistent level. Its range of application includes acute and chronic pain associated with sports injury. Furthermore, the portable nature of PainShield® enables patients to use the device at home, work, or in a clinical setting. If you’re strapped on time, PainShield® can also be used while you’re asleep. Overall, NanoVibronix PainShield® is intended to be an accessible product capable of providing a wide range of benefits including faster recovery time, high compliance, and increased safety and efficacy over existing ultrasound devices.
“The pain relief market is flooded with products that simply mask the pain versus providing proven pain resolution. We believe the opportunities are endless with this product. Our initial entry, as well as our next generation, which is currently in development…Our entry into the OTC market will be preceded by submission of the final results of our Lateral Epicondylitis (Tennis Elbow) study,” continued Brian Murphy.
NanoVibronix has submitted its trademark application to the U.S. Patent and Trademark Office (USPTO) for protection of PainShield RELIEF™ and expects to submit for clearance to the U.S. Food and Drug Administration (FDA) in the near term. As someone who suffers from tennis elbow, I would happily invest in a PainShield® device if it ever became available for OTC in Ontario. As for investors, keep an eye out for the Company’s Lateral Epicondylitis study utilizing PainShield®. For more details regarding NanoVibronix latest news and financials, check out this article.
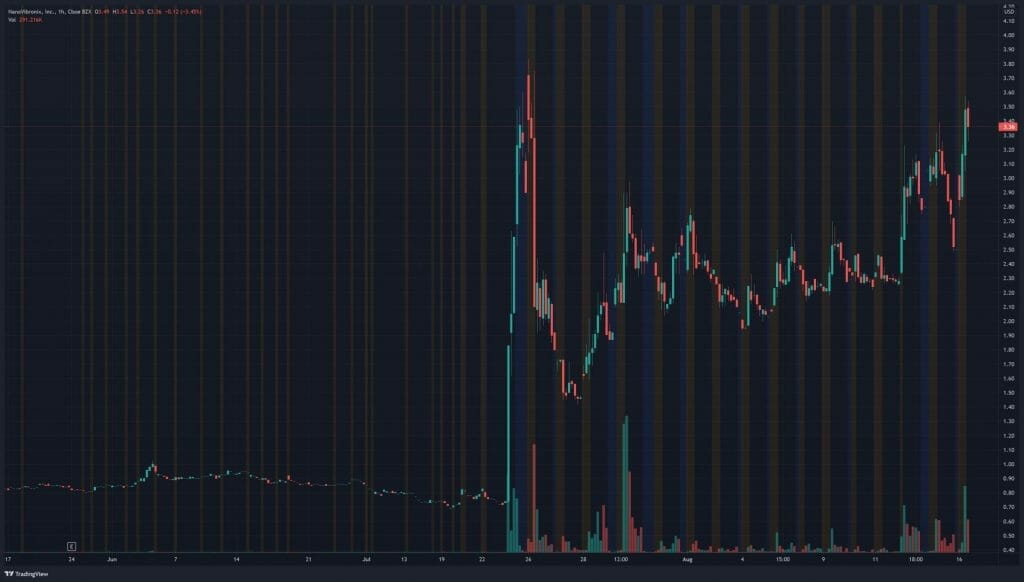
NanoVibronix’s share price opened at $3.18, up from a previous close of $2.52. The Company’s shares are up more than 33% and are currently trading at $3.38 as of 10:31 AM ET.


