Mindset Pharma (MSET.C) announced their lead clinical candidate, MSP-1014.
MSP-1014 is a differentiated psilocybin-based compound from Mindset’s Family 1 of novel, patent-pending psychedelic compounds. MSET plans on moving forward into current good manufacturing practice (cGMP) compliant manufacturing and IND-enabling studies.
“The selection of Mindset’s first lead candidate, MSP-1014, is a significant milestone in our clinical journey. We are excited to enter the final preclinical development step prior to commencing first-in-human clinical trials. Our next generation psychedelic compounds represent the flourishing evolution of therapeutics to effectively address neurological and neuropsychiatric disorders,” stated James Lanthier, CEO of Mindset.
“MSP-1014 demonstrates superior preclinical characteristics in head-to-head comparison with psilocybin and its active metabolite psilocin, including increased safety and efficacy, which we believe will potentially result in lower clinical doses thereby indirectly decreasing safety concerns, and manufacturing advantages. MSP-1014 has the potential to be a safer, more efficacious analog to psilocybin, with reduced potential side effects. Given its chemical profile, we anticipate that MSP-1014 will have the potential to treat mood disorders, including major depressive disorder, substance misuse disorders and end-of-life angst associated with terminal illnesses, including cancer. Therefore, we believe that MSP-1014 has the potential to be a first-in-class psychedelic drug candidate. With our first lead candidate selected, Mindset continues to steadily advance its position in developing innovative next-generation psychedelic therapeutics that couple the life-changing potential of psychedelic drugs with novel and patentable new chemical entities that are more predictable, convenient, and safer than first-generation psychedelics.”
“Given its chemical profile, we anticipate that MSP-1014 will have the potential to treat mood disorders, including major depressive disorder, substance misuse disorders and end-of-life angst associated with terminal illnesses, including cancer. Therefore, we believe that MSP-1014 has the potential to be a first-in-class psychedelic drug candidate.”
As Lanthier stated above, Mindset believes that MSP-1014 has the potential to depression, substance abuse, and end-of-life-angst. That last one stands out to me, because not many psilocybin companies talk about it. We currently have treatments for depression and substance abuse, and yes, you can definitely argue about the inadequacy of those methods, but they exist, and many people get excited when psilocybin is proven to be as effective as those existing methods.
But currently, there are very few – if any – effective end-of-life treatments. Michael Pollen’s popular book How to Change Your Mind includes the story of Patrick Mettes, a 53-year-old who was dying of cancer, who took psilocybin in order to deal with end-of-life anxiety. The results were profound: he was able to quell his fear of death, and lived out his remaining 13 months with a purpose, rather than living paralyzed with death anxiety.
Because currently the treatment for death anxiety is pretty much just ‘figure it out for yourself’, it’s hard to estimate an exact dollar market for this kind of treatment, but it’s worth noting that every year in the US ~600,000 people die from cancer.
Mindset selected MSP-1014 because of its strong safety profile from their preclinical studies, and it has undergone a comprehensive range of in vivo and in vitro tests. Mindset intends to further develop candidates from Mindset Families 2-4 with the goal of selecting additional lead candidates in the near future.
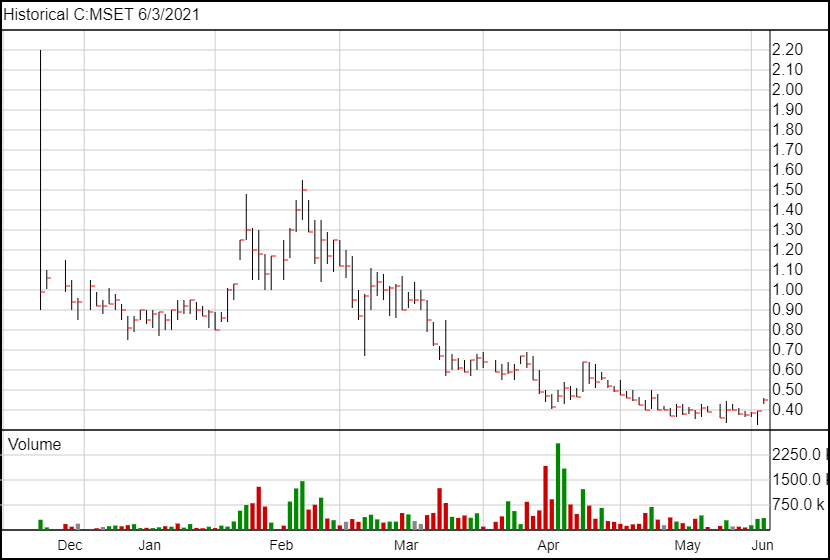
Following the news, Mindset’s share price is up 5 and a half cents and is currently trading at $0.45.