Mindset Pharma (MSET.C) announced they have filed a new patent with the US Patent and Trade Office (USPTO), extending its Family 4 compounds, a set of novel DMT/5-MeO-DMT analogs.
Mindset’s Family 4 compounds are DMT and 5-MeO-DMT inspired psychedelics that offer a broad range of pharmacological diversity suitable for in-clinic settings. DMT and 5-MeO-DMT are seen as advantageous because of the shorter trip time, which typically lasts 15-30 minutes, compared to psilocybin, which can last up to 8 hours.
Mindset are one of the first groups to file patent applications on next-generation versions of DMT and 5-MeO-DMT. According to the CEO of Mindset, James Lanthier, the goal is to make their version of DMT and 5-MeO-DMT “safer, more predictable, [and] more effective.”
MSET filed their first DMT and 5-MeO-DMT patent back in March, noting that the “5-MeO-DMT and DMT’s potency at the 5HT1A receptor in addition to the 5HT2A receptor is unique compared to psilocybin and further increases the breadth of pharmacological diversity of Mindset’s proprietary compounds.” In this newest patent, Mindset says they are applying lessons from the more than 80 psychedelics they have developed.
“The second patent application for our novel drug candidates incorporates new side-chain restricted analog designs – working from the progress of our first application – and broadens the strength of our patent estate,” stated Lanthier. “Our DMT/5-MeO-DMT analogs have shown a range of duration and pharmacological diversity in preclinical modeling, with the potential to offer a more convenient and individualized https://e4njohordzs.exactdn.com/wp-content/uploads/2021/10/tnw8sVO3j-2.pngistration for clinic-based therapies.”
Mindset has four psychedelic families. Family 1 compounds focus on using design strategy to improve the safety profile of their psilocybin and psilocin compounds. Family 2 compounds are Mindset’s next-generation psilocybin and psilocin drug candidates optimized for in-clinic settings. Family 3 is their microdosing family, which combines longer action time with lower doses to allow people to go on with their regular activities while using the substance.
Today’s application was Mindset’s seventh patent application. The above-mentioned first DMT application in March was their sixth, so DMT and 5-MeO-DMT have been the focus of their patent efforts recently. However, Mindset’s planned clinical trials for 2022 are for the Family 1 psilocybin-inspired MSP-1014, which they selected as a lead candidate in June.
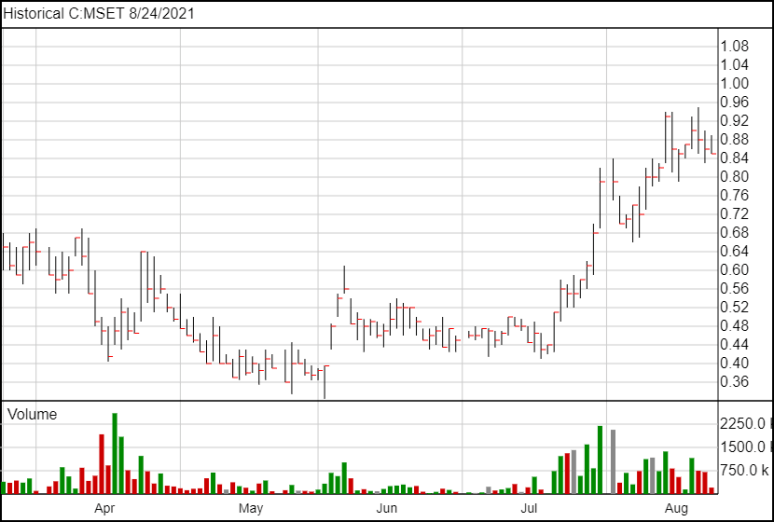
Following the news, Mindset’s shares are down 1 cent and are currently trading at $0.85.