Esketamine
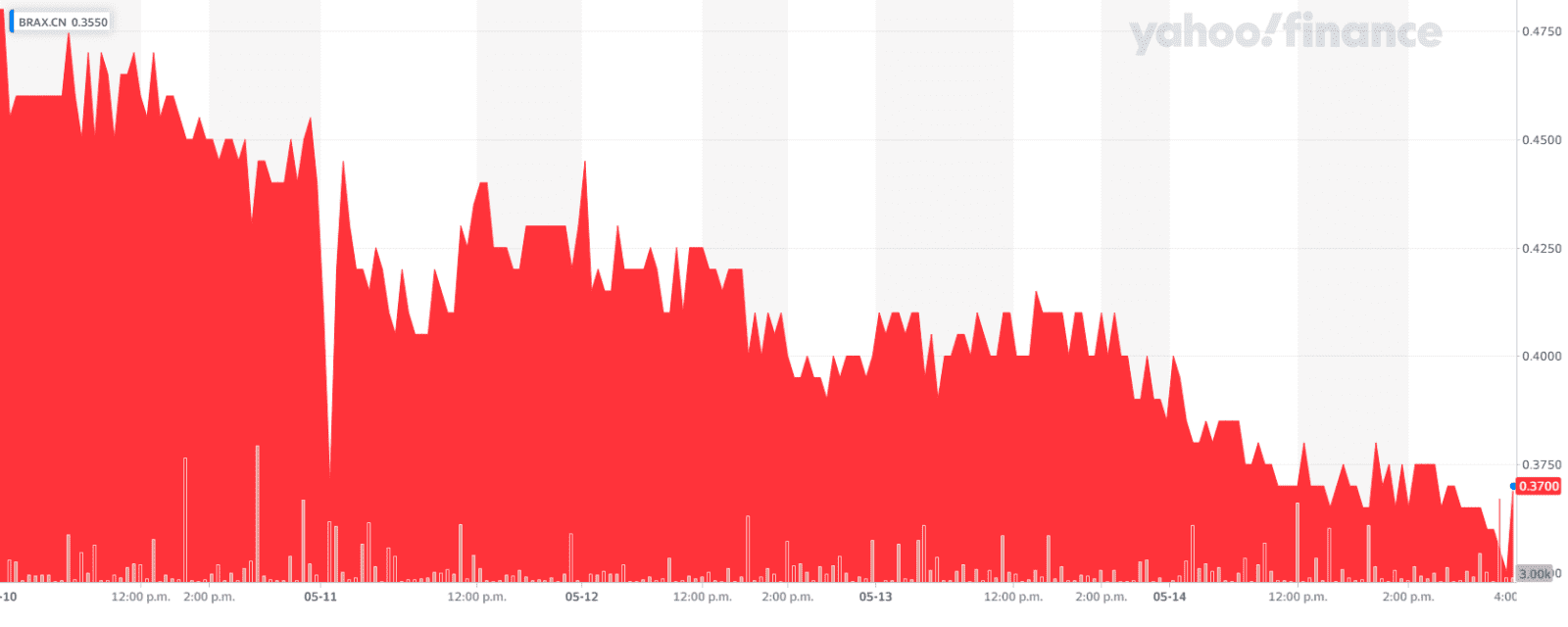
Braxia Scientific (BRAX.C), formerly known as Champignon Brands had a rough 2020.
It was a down year for many of us, but Champignon really found itself in trouble after its former CFO messed up their financials so badly he missed the asset calculations by about $13 million CAD, and then to add insult to injury filed several documents late. He has since been let go by the company and was paid only $7,500 CAD in salary on top of 300,000 options, but damn did he set the company back a ways. This lead to a lengthy frozen stock, followed by a class-action lawsuit from investors.
Overall, not a fun time. I wrote about their previous debacle and actually gave the company the benefit of the doubt. Braxia is now moving forward, with its trading again on the CSE.
Braxia’s first rumblings back in 2019 were around mushroom teas and supplements. The company pivoting from mushroom powders and teas to ketamine clinics was a bit out of left field in terms of a business plan, but, businesses need to adapt. And the ketamine clinics have been very profitable, just look at Field Trip (FTRP.C) and Novamind (NM.C) who are doing millions in revenue from a handful of clinics. And whatever about the mushroom powder, 800,000 companies are already doing it, onto bigger and better things.
The ketamine clinics found a bit of a loophole -esketamine, a ketamine derivative that doesn’t come with all of the red tape of pure ketamine. Esketamine is also legal, and approved for use by Health Canada.
This synthetic ketamine is similar to synthetic psilocybin companies like Braxia, Cybin (CYBN.NE), Mindset Pharma (MSET.C), Atai, and Compass Pathways (CMPS.Q) are all creating. Currently, the main buyers of synthetic psilocybin are companies and universities doing pre-clinical and clinical studies on psychedelics. Synthetic psilocybin is required for most of these studies. It’s not a huge market, but it’s something to generate early revenue.
Quebec
Braxia announced earlier this week it was opening the first ketamine clinic in Quebec by entering a partnership with Neurotherapy Montreal. This will be the company’s fourth clinic it’s opened so far. Neurotherapy Montreal’s large, established patient base includes patients diagnosed with brain-based injuries such as Anxiety, Depression, Attention Deficit Hyperactivity Disorder (ADHD), Traumatic Brain Injury, Stroke, Multiple Sclerosis, Parkinson’s Disease, Alzheimer’s Disease, Chronic Pain, Vestibular and Sleep Disorders.
[youtube https://www.youtube.com/watch?v=Zc6sHqm_74s]
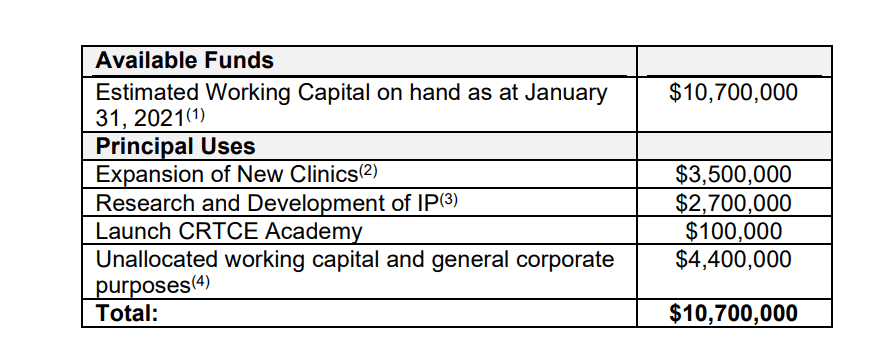
Braxia is planning to scale its clinic business and bolster its R&D for the purposes of building IP. In August 2020, the US FDA approved an esketamine nasal spray as a treatment for depression with suicidality. Ketamine may also be used in combination with other commonly prescribed antidepressants for the treatment of major depressive disorder in adults who have not responded to at least two antidepressants.
Braxia is preparing provisional patents for their ketamine and psilocybin derivatives, which it expects to submit in mid-2021 before it seeks to synthesize them at a subcontracted lab.
Braxia is also focused on utilizing its own clinical research ecosystem for its psilocybin trial, a Phase 2 clinical trial involving adults with treatment-resistant depression. Braxia will be collaborating with the Brain and Cognition Discovery Foundation.
Useful subsidiaries
Braxia has a handful of subsidiaries that operate its multiple divisions like R&D/IP development, clinical studies and ketamine clinics.
Through Braxia’s wholly-owned subsidiary, CRTCE, Braxia is looking to create a Canadian-based broad network of specialty clinics for ketamine treatment and psychedelic-enhanced psychotherapy to help patients with depression, anxiety, and addiction, all of which are on the rise in the COVID era. Currently, there are six affiliated psychiatrists, and 50 consulting staff working at the CRTCE Clinics, including anesthesiologists, registered nurses, pharmacists, and researchers.
In addition to its clinics, CRTCE is planning to implement an academy (the “CRTCE Academy”) to train healthcare providers on the implementation of ketamine and related psychedelics for adults with common mental health conditions. Once implemented, the CRTCE Academy is expected to train healthcare providers on pharmacology and integrated psychosocial/psychotherapeutic interventions that are evidence-based and, where required, approved by regulators.
Braxia acquired the biotech company Novo Formulations in March 2020 for IP development. Novo’s IP’s scope focuses on novel and innovative delivery systems for the pharmaceutical industry. Novo is actively formulating and developing bioavailable, delivery platforms, including topical creams, intranasal spray, and sublingual tabs.
Novo is also developing an intranasal formulation of racemic ketamine for the treatment of depression and pain. Chemical and analytic stability tests are being conducted at McMaster University. The project is set to be completed sometime later this year.
https://equity.guru/2021/05/10/the-results-are-in-for-maps-phase-3-study-of-mdma-assisted-therapy-for-severe-ptsd/
Last year Braxia acquired Tassili for clinical research. Tassili has a collaborative research agreement with the University of Miami’s Miller School of Medicine. The agreement covers dosing rats and mice who have been afflicted with a post-traumatic stress disorder or traumatic brain injury with PTSD a combination of psilocybin and CBD. The objective of the Tassili pre-clinical trials is to assess how the combination of psilocybin and CBD may mitigate the adverse effects of PTSD and a traumatic brain injury with PTSD.
Braxia also purchased the biotech company Altmed last year. The transaction gave Braxia three trials in the Phase I stage and three trials in the preclinical stage during 2020, as well as seven patents for its ketamine/psilocybin delivery platforms and formulations.
Upside
Braxia has its hand in a lot of different areas. The company’s reputation will take a while to restore, but for the time being, the stock is cheap. If ketamine clinics are the way of the future and you are looking for an earlier stage Field Trip with a small market cap this might be it.
https://equity.guru/2021/03/17/ketamine-clinics-proving-to-be-significant-revenue-generators-for-early-stage-psychedelics-companies/
In terms of profitability, the ketamine clinics seem to be where it’s at. I would bet more and more companies will start to acquire them as it’s also a great way to begin building a patient database for the future. Companies that already have a list of clientele will be in a stronger position if new and novel drugs and therapies are developed, or federal regulations around psychedelics change.
By building up customers Braxia is putting itself in a great position to grow in the coming years.
Hopefully no more late filings.






$5 CAD 1Year target