AnPac Bio-Medical (ANPC.Q) announced today that it has been granted its first disease treatment patent by the United States Patent and Trademark Office. Additionally, the Company also announced that its newly launched cancer treatment joint venture has secured over USD$4 million of capital contributions from multiple funding sources.
“We are pleased that AnPac Bio has been granted our first disease treatment patent, whose disease treatment related claims will be licensed to our new cancer treatment joint venture (while future disease treatment only patent applications will be transferred to the cancer treatment joint venture). We are also pleased that our cancer treatment joint venture has secured funding to speed up its cancer treatment developments. Our core business of cancer detection and this new cancer treatment venture are expected to have significant synergies in terms of technology, market, business and customer,” said Dr. Chris Yu, Chairman and CEO of AnPac Bio.
AnPac Bio is a biotechnology company focused on pre-cancer and cancer screening to develop early disease detection devices that are more accessible. With this in mind, AnPac Bio has developed a new way to detect cancer and pre-cancer through its Cancer Differentiation Analysis (CDA) device. The CDA device uses an integrated sensor system to detect several biophysical signals. The Company’s proprietary CDA algorithms are then able to synthesize this data to generate a personalized risk assessment for each patient.
Representing the twenty first patent granted to AnPac Bio in the United States, the Company’s latest patent will cover novel device structures and methods to modify biological samples for treatment purposes. As previously mentioned, AnPac Bio has also launched its cancer treatment joint venture, which secured more than $4 million in funding. These funds will be used towards the fabrication of cancer treatment medical devices and the devices’ subsequent laboratory tests this year.
The new cancer treatment technology is expected to be based on AnPac Bio’s finding in the past 10 years of extensive early-stage cancer detection work involving over 222,200 samples. This includes commercial CDA-based tests and CDA-based tests for research purposes. The aim of this technology is to prevent disease progression for both pre-cancer and cancer patients.
“AnPac Bio has always focused on technological innovation and building a strong patent and IP portfolio in cancer screening and detection. We will continue to invest aggressively in advanced cancer detection and treatment technologies and related intellectual property, and maintain our competitiveness in cancer detection…“AnPac Bio will continue to focus on its core business cancer screening while supporting the development of the new cancer treatment joint venture,” continued Dr. Chris Yu.
AnPac expects that laboratory tests of this technology will begin in Q3 2021. Additionally, a class III medical device registration application to the Chinese Medical Products Administration (NMPA) is planned for Q3 2022.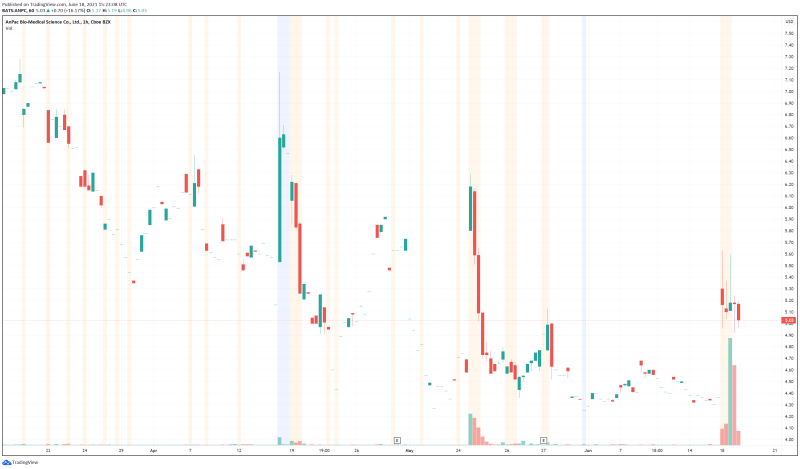
AnPac Bio’s share price opened at $5.28, up from a previous close of $4.33. The Company’s shares are up 16.63% and are currently trading at $5.06 as of 11:24AM ET. This indicates that there has been significant change following the news.

