Revive Therapeutic (RVV.C) announced they have entered into an agreement with the University of Health Sciences Antigua (UHSA) to collaborate with Revive on their novel psychedelic-assisted therapies and pioneering the clinical research and development of psychedelics in Antigua and Barbuda.
The collaboration has a number of goals, including the establishment of a psychedelic treatment center in Antigua and Barbuda using Revive’s novel psychedelic formulations and delivery systems. This will include Revive’s proprietary oral-thin film (OTF) delivery system, which dissolves in water in less than five minutes, with dosages between 1 mg and 20 mg. OFT delivery allows better dosage control and can deliver the psilocybin faster, as well as helping those who have trouble swallowing take the drug.
The collaboration has a number of goals, including the establishment of a psychedelic treatment centre in Antigua and Barbuda using Revive’s novel psychedelic formulations and delivery systems.
The partnership will lead to the establishment of a Master’s in Psychedelic Medicine for Doctor of Medicine students at UHSA, allowing students to gain more knowledge and experience in the field. As part of the collaboration, UHSA will exclusively use Revive’s intellectual property for research, and eventually Revive hopes that this will provide a foundation for further novel psychedelic therapy research and commercial use in Antigua and Barbuda.
“Through our partnership with Revive Therapeutics, we have the unique opportunity to become a regional leader in the research of psychoactive therapies for mental health. Our medical students will now have access to the scientific knowledge and uses of treatments long before joining the physician workforce, which is a great benefit. We too are excited to partner with Revive as we contribute to the future of medicine,” commented Dr. Adedayo Akande, President of UHSA.
Revive also has hopes that this partnership will help accelerate their US FDA clinical development plans.
Revive also hopes that UHSA’s research will help them support regulatory drug approvals in the Caribbean, which often have more relaxed laws surrounding psychedelics than us mainlanders do. Revive also has hopes that this partnership will help accelerate their US FDA clinical development plans.
Any good research, done anywhere, helps a company understand their product better, so partnering with UHSA will provide Revive with valuable information surrounding their psilocybin product which they can use when they’re seeking regulatory approval in the future.
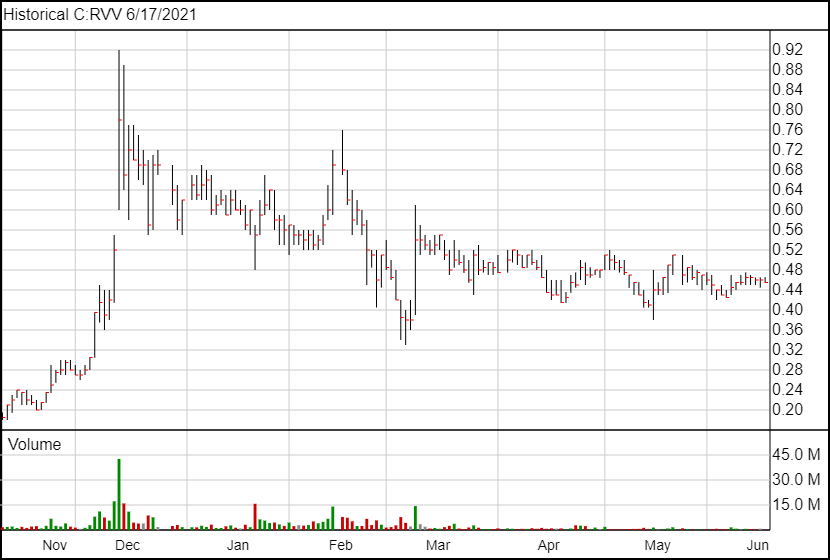
Following today’s news, Revive’s share price has not changed.
Full disclosure: Revive Therapeutics is an Equity Guru marketing client.