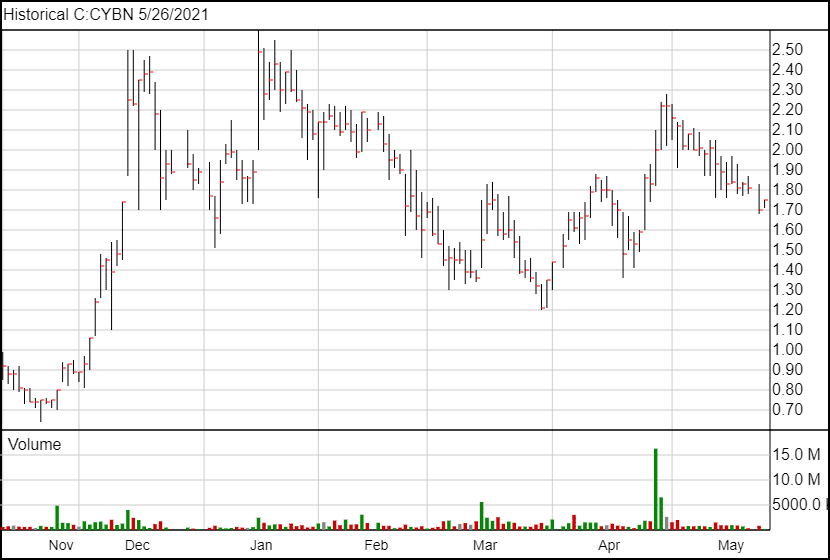
Cybin (CYBN.NE) announced they have filed an international patent application that brings the potential to obtain patent coverage in 153 countries.
The application is governed by the Patent Cooperation Treaty (PCT) and grants Cybin the right to file future national applications into treaty member jurisdictions. According to Cybin’s press release, PCT claims a library of psychedelic derivative drug development candidates.
According to the World Intellectual Property Organization, the PCT “assists applicants in seeking patent protection internationally for their inventions, helps patent offices with their patent granting decisions, and facilitates public access to a wealth of technical information relating to those inventions.”
The list of countries that are party to the PCT include Canada, the US, China, Brazil, India, all of Europe, and most other countries in the world (even North Korea). Countries not party to the deal include Venezuela, Argentina, and the DRC.
Cybin re-iterated that they continue their three-pillar drug development program, and added that this PCT application will allow them key intellectual property protection internationally. The three pillars of their program are (1) novel drug discovery and research on the potential efficacy of psychedelic molecules to address unmet mental health needs, (2) efficient drug delivery for enhanced dosing control, and (3) a novel treatment program.
“Technologies gained by Cybin resulting from our strategic acquisition of Adelia Therapeutics, coupled with subsequent research initiatives from our experienced scientific team, have produced Cybin’s first PCT filing. As we progress our R&D and clinical programs, we expect to continue to create and develop innovative therapeutics with faster onset of action, smoother pharmacokinetic profiles, shorter treatment periods, and reduced side effects,” stated Doug Drysdale, Chief Executive Officer of Cybin.
By filing an application with PCT, Cybin is showing confidence in their ability to reach new jurisdictions. Because countries have different laws regarding the legality of psychedelic drugs, having an easier way to file patents across the world will allow them to take advantage of differing laws across the world.
For instance, if they decided to go down this route, a PCT patent could help them in the psychedelic tourism business. Even if they don’t want to engage in psychedelic tourism, international patents could help them take advantage of less restrictive laws for lab and clinical research abroad. They are already doing the latter by conducting the CYB001 Phase II study in Jamaica.
Following the news, Cybin’s share price is up 3 cents and is currently trading at $1.73.
Full disclosure: Cybin is an Equity Guru marketing client.