Humming in Q1
I’ve had a rough year, but Revive (RVV.C) sure hasn’t.
With a portfolio based around psychedelics, cannabis, and diseases – this company has made 3 swishes in the current market landscape.
Today they announced the closing of their PharmaTher purchase, making their portfolio even more diverse. For $10M CAD they acquired:
- All intellectual and work property derived from pre-clinical research activities from the National Health Research Institutes (“NHRI”) in traumatic brain injury and stroke, as it relates to psilocybin with the aim to obtain FDA Orphan Drug Designation.
- Psilocybin in the Treatment of Neurological Brain Injury – Relates to pharmaceutical compositions comprising psilocybin and their use for the treatment of neurological brain injuries and migraines.
- Use of Psilocybin in the Treatment of Cancer – Psilocybin’s use of significant unmet medical needs for Liver Carcinoma, Melanoma, Breast Neoplasms, Kidney Neoplasms and Acute Myeloid Leukemia.
- Psilocybin Pharmaceutical Combination Therapies – Novel combinations of certain FDA approved drugs with psilocybin as a potential therapeutic option to reduce the side effects and improve the effectiveness of psilocybin to treat neurological disorders.
It’s been a big month for Revive.
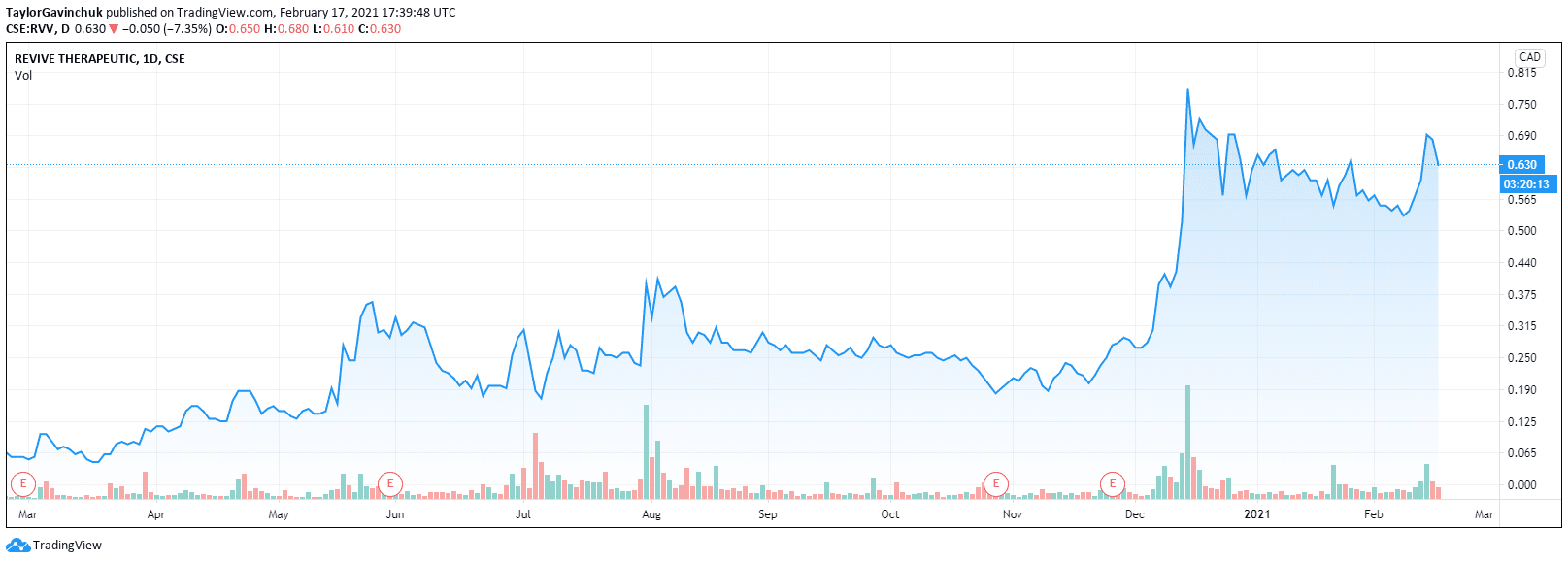
On February 12th, the company closed its oversubscribed $23M CAD financing at $0.50 per unit, backed by Cannacord.
Revive will use the cash for its phase 3 clinical costs for Bucillamine for COVID-19, phase 1 clinical costs for Psilocybin for methamphetamine use disorder study, and other psychedelic drug formulation development work.
Patents
Along with clinical trials, Revive will also be creating its own psilocybin through its research partner NC State University.
Revive seeks to develop and commercialize its own pharmaceutical-grade psilocybin with this biosynthesis technology at scale to support the Company’s current psilocybin-based product pipeline that includes an oral thin film product currently being developed in collaboration with the University of Wisconsin-Madison and the intellectual property and research with psilocybin being developed by PharmaTher Inc.
Earlier this week I wrote about a debate brewing in the psychedelics space between synthetic and natural psychedelics substances. Long story short, while many people will take natural products over synthetic ones, a more important point for investors is, however – psilocybin as a compound is not patentable.
Revive is making a bet that their patents which include delivery methods and future products will hold value.
The most interesting thing for me about Revive’s patents is they cut across both the medical and recreation side of psychedelics as you have gumdrops, as well as purely pharmaceutical delivery methods.
Revive Therapeutics (RVV.C) inks research agreement with NC State for synthetic psilocybin
They have an impressive list of patents filed, which could be extremely valuable in the long term.
Some of these include:
- Use of Bucillamine in the Treatment of Infectious Diseases, including COVID-19
- Psilocybin effervescent and psilocybin tablet – Solid Oral Pharmaceutical Compositions
- Psilocybin hard-shell capsules – Pharmaceutical Capsule Compositions
- Psilocybin gum drops – Pharmaceutical Gumdrop Compositions
- Psilocybin oral strips and transmucosal – Thin-Film Pharmaceutical Delivery System and Formulations
- Use of Cannabidiol in the Treatment of Autoimmune Hepatitis
https://equity.guru/2020/10/21/havn-life-sciences-havn-c-supplies-mushrooms-for-revive-therapeutics-rvv-c-clinical-trials/
COVID19
The company is positioned for psychedelics, cannabis, and diseases (including COVID 19), all three of which can explain the stocks increase over the last year. Their study into Bucillamine as a treatment for COVID 19 reached phase 3 last year, the trial will see up to 1,000 participants for 14 days.
The study will be looking at COVID patients with harsh conditions like a fever (oral temperature ≥38°C), cough, shortness of breath, chest x-ray changes consistent with COVID-19 at the time of screening.
Bucillamine has a well-known safety profile and has been prescribed in the treatment of rheumatoid arthritis in Japan and South Korea for over 30 years.
Preclinical and clinical studies have demonstrated that reactive oxygen species contribute to the destruction and programmed cell death of pulmonary epithelial cells. N-acetyl-cysteine (NAC) has been shown to significantly attenuate clinical symptoms in respiratory viral infections in animals and humans, primarily via donation of thiols to increase the antioxidant activity of cellular glutathione.
The stock took a big leap once the company received FDA approval.
https://equity.guru/2020/07/31/revive-therapeutics-rvv-c-blasts-off-68-fda-phase-3-covid-19-approval/
A sleeper
Coming in at a $119.3M CAD market cap, Revive is a nice size right now.
They aren’t massive like a Compass Pathways ($1.74B CAD) or MindMed ($1.69B), but they aren’t a risky distant speculation either. According to Psilocybinalpha, Revive has the 6th highest market cap of the 30ish psychedelics pubcos.
Revive was also picked up by Horizon’s new psychedelics ETF.
Revive Therapeutics (RVV.C) joins the first North American psychedelics ETF
Revive is one of those ‘sleeper’ companies in the psychedelics space that potentially have a lot of groundbreaking press releases coming their way over the following months and years.
Psychedelic therapy studies have been fast-tracked by the FDA, and with the team of scientists and doctors on Revive’s team, along with their already existed clinical trials this company looks well-positioned to take on the complicated psychedelics industry.
Revive could be a nice addition to a portfolio as it sits just on the edge of mid/large-cap with respect to the psychedelics space.
The way the company has set up its product pipeline and patents, they have a massive amount of kicks at the can, and with the smart moves this company has made in the past, it would be surprising if at least one of these avenues doesn’t pan out for investors.
Revive has a lot of room to run with a lot of favorable news releases potentially coming its way.
Full disclosure: Revive Therapeutics is an equity.guru marketing client.