Pure Extracts Technologies (PULL.C) set their milestone timetable for their study on both the formulation and manufacturing of psilocybin-based treatments today, according to a press release.
The study’s focus will be on dosage testing for formulating, manufacturing and bioavailability that could be used in any future clinical trials by both Pure and their pharmaceutical customers. The company has put together clinical batch manufacturing, packaging and labelling into the study, in addition to conformity testing and stability testing. The study will conform to both Canadian Good Manufacturing Practice (GMP) and Good Clinical Practice Standards (GCP).
“We are excited about the R&D progress we are going to make with TIPT over the next 6 – 8 months while waiting to receive our Dealer’s License from Health Canada. We will be well prepared for our move into the controlled substances world of psychedelics and will have advanced knowledge regarding psilocybin and its associated novel delivery mechanisms,” said Ben Niklaevsky, CEO of Pure Extracts.
The key milestones in the 250-day timeline include:
- Sourcing and procurement of medicinal ingredients, excipients & packaging materials
- Raw Material Testing
- Formulation and Manufacturing Process Development, Stability Testing
- Clinical Batch Manufacturing
- Quality Control Release Testing
- Shipping of Clinical Batches to CRO
The company is intending on producing these formulations in an oral tablet, capsule and nasal gel format, which will be used as an investigational product and overseen at the Toronto Institute of Pharmaceutical Technology by Dr. Alexander MacGregor, who has been doing the science duties for the company.
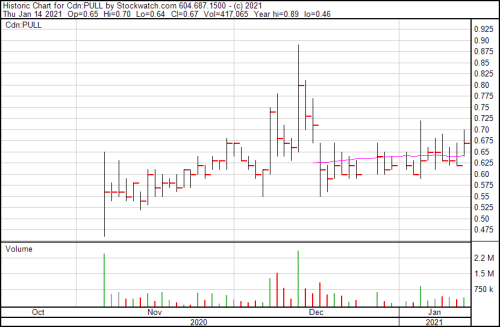
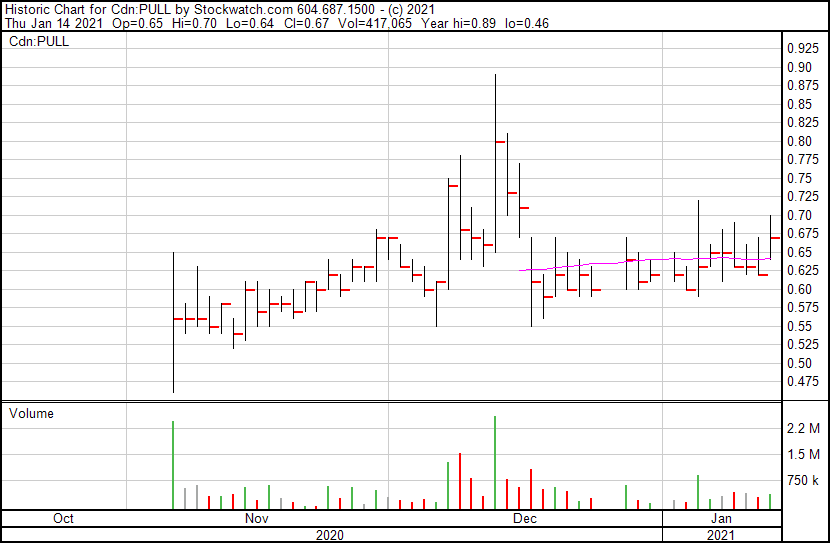
PULL is up 8.1% and now trading at $0.67
—Joseph Morton
Full disclosure: Pure Extracts Technologies is an equity.guru marketing client.