For the last several years, a lot of stories I’ve written on this site have been about promise. The promise of something better, cheaper, with more upside, with less downside. Promise that a team will do more, or do better, or do something – anything. Promise that, if the planets align, something big will come.
Sometimes that promise comes close. Sometimes it doesn’t come anywhere near. Sometimes, as we’ve seen with recent stories in the nickel business, it’s a scam from the outset.
I think a lot of us have come to a point in these markets where promise isn’t so exciting as it once was. A point where we fondly recall words like ‘dividends’ and ‘profit’, and where we look for companies where the risk has been limited, and replaced with actual reward. Where questions are asked about ‘growth’ rather than ‘runway’ and ‘burn.’
In the cannabis world, after several years of stupid-hype, the single dose of adulting came through a company called Cannaroyalty, more recently Origin House (OH.C), which was acquired by Cresco Labs (CL.C) for a nice fat profit. In the medical world, Well Health (WELL.C) has done amazing things rolling up clinic chains and fast tracking their technological advancement and revenue growth.
The key to those plays wasn’t to promise they’d be the next billion dollar public company and compel everyone to ‘come and ride the train, choo-choo ride it.’ The key was to not overpay for assets, to buy into things that were complimentary to one another instead of just market cap exploders, to receive a big enough piece of those assets that they had a say in their upgrading and growth, and to become bigger by factors based on how well those assets actually worked together.
Yesterday, Aurora Cannabis (ACB.T), which went the other way, of raising way too much money to pay way too much for things they ultimately couldn’t use, announced financials that saw a net loss of $33 per $7 share. Billions written off. Revenues down. It’s a mess of gargantuan proportions, even moreso because most cannabis pubcos tried to chase them off the cliff they threw themselves at.
Elsewhere, a tiny biotech company is going the other way.
I only know about it because Marc Lustig, he who built Origin House from a few million in seed capital to a billionaire’s exit, told me he really liked it, and especially liked the team. I know Lustig well enough that he doesn’t waste time with dreamers – he likes real assets, real revenue, real plans that are executable without needing the laws to change, and teams packed with guys who’ve done it before.
Lustig bought into more than 25 cannabis assets to build Origin House, and flipped many more for a profit. Not ‘take your pubco to a 5x’ profit, but consistent ‘show your investors you’re real and trustworthy’ profits that made it easy for him to buy more, and for investors to hang in for the long haul.
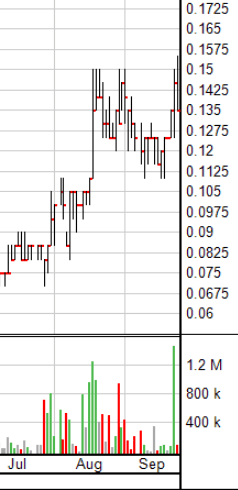
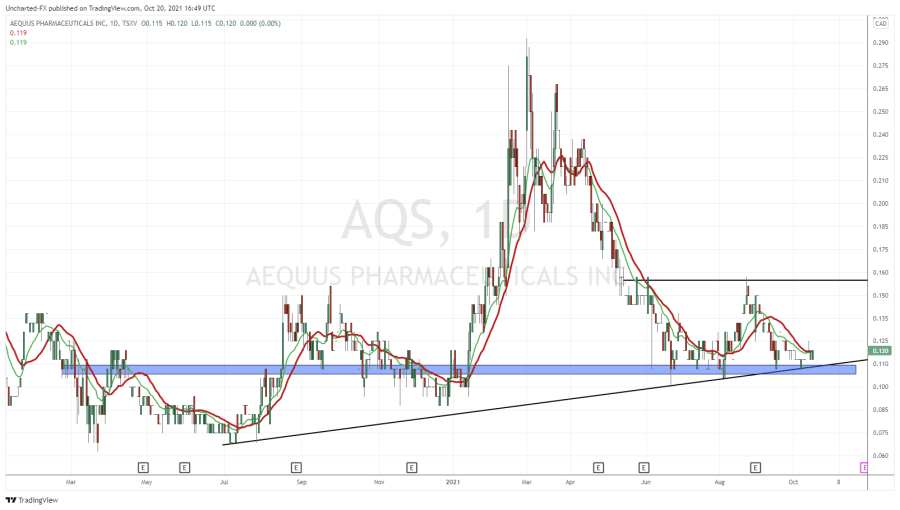
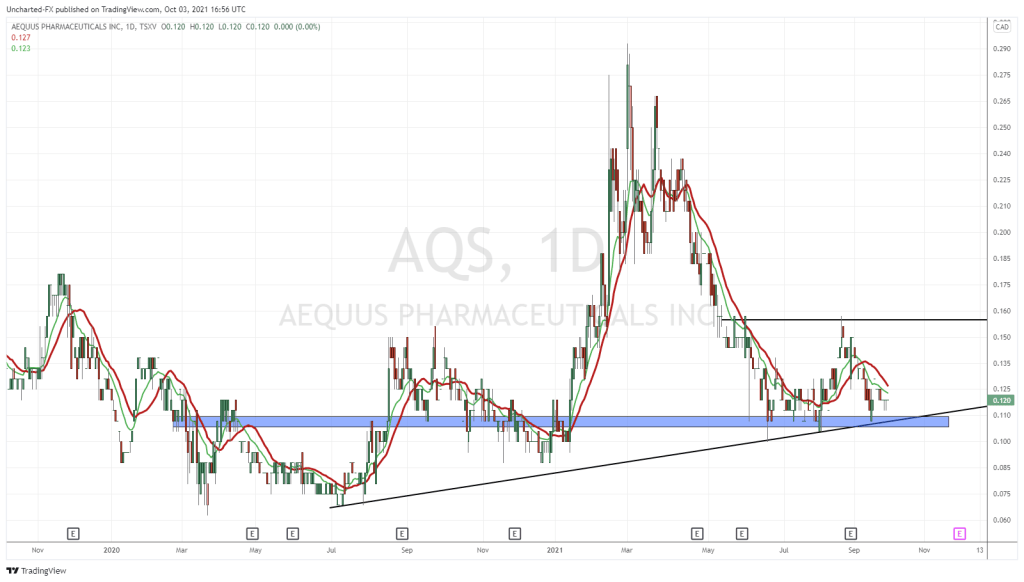
From what I can see of Aequus, and from talks with their team in the last several weeks, AQS is built from similar stone.
The big difference between AQS and any other Canadian biotech company for me is confirmed with this paragraph from their just dropped news.
Aequus has now submitted three new products for Health Canada approval. As with the previous submissions, the additional Evolve product was submitted for approval with Health Canada as a Class II medical device. According to Health Canada, the target review time for a Class II MDL application is typically 20 days.
20 days.
No five-year FDA trials, where hundreds of millions need to be spent on research and submissions and animal tests and human tests and clinical tests, and where every step runs the risk of an unexpected hurdle that can add a year to the approval timeline.
No, AQS is taking existing APPROVED products that have already gone through the wringer and made it to shelves, and stacks them together, or alters delivery platforms, or improves their accessibility in small ways that, yes, do need an okay from the regulators, but where that okay is a mere bureaucratic process that must be undertaken, and not a capital intensive multi-year lottery ticket.
Don’t get me wrong, there’s definitely big money in finding something that needs approval and getting it some. But even the Pfizers and Glaxos of the world are playing the lottery when they bring something new – they just have the size to absorb the failures.
Generally, small biotech companies don’t. They go all in on one product and investors stand next to them yelling, “No whammies!” for three to five years.
AQS isn’t playing that game. They find something that is approved, works, and has been selling already, and with which their team suspects they have an idea of how to make that product better. They buy the rights to that IP for a fair amount, but nothing absurd, and make the necessary submissions to health officials to get the changes approved – quickly and cheaply.
As an example: A medication that is currently approved as a suppository, might be easier to sell if you could deliver it with a nasal spray, or a patch. Or a drug that plays havoc on the digestive system may be better off bypassing the digestive system entirely by being used as a cream instead of a pill.
I’m not a scientist, but this makes a lot of sense to me, and some fairly large investors besides me, so I’m into where Aequus wants to be.
As I write this story, Aequus has announced a third product it’s moving forward with, an additional product to go along with the two other ‘dry eye’ treatments they’re currently submitting to Health Canada as a Class II medical device.
“In preparation for launch, we have engaged Canadian eye care professionals by creating advisory boards and research groups, built B2B relationships with clinic networks, added specialized personnel to our commercial team, and planned branded marketing campaigns to targeted customer segments,” said Doug Janzen, chief executive officer and chairman of Aequus Pharmaceuticals. “Our team has a deep understanding of ophthalmology and years of operational experience. We see our new product launches as being the perfect fit for Canadian patients who have been wanting a wider range of options for dry eye treatments.”
- Simple.
- Talking to medical professionals. Check.
- Create your advisory board. Check.
- Go to existing clinics and businesses that have deep ophthalmology patient databases. Check
- Market specifically to that small group, which supplies treatment to a much larger group. Check.
The products AQS has under its umbrella ALREADY SELL TO CONSUMERS. That means there’s revenue afoot.
That revenue isn’t going to spin your head off, at least not yet. It’s not going to be a case of financials blasting out and stock prices jumping 30% every second Tuesday. This won’t be a day-trader darling.
But that’s kind of the point. AQS will be a nice little earner that builds over time, where the pieces that are acquired are either upgraded cheaply to bring a wider consumer base, or re-submitted to work better with the other acquired pieces – or both – and where the major risk for the investor is, one day we wake up to find dry eyes cease to be a problem.
Right now, with the markets either stupid up or stupid down, depending on what an insane President has to say on a given day or how much printer ink the mint has left, I want stable, revenue earning, scale-able companies where the cash on hand isn’t far from the market cap of the company proper.
Aequus plans to build on its Canadian commercial platform through the launch of additional products that are either created internally or brought in through an acquisition or licence, remaining focused on highly specialized therapeutic areas.
People are figuring it out.
I’m in.
— Chris Parry
FULL DISCLOSURE: Aequus Pharmaceuticals is an Equity.Guru marketing client, and we’ve bought stock in the company.